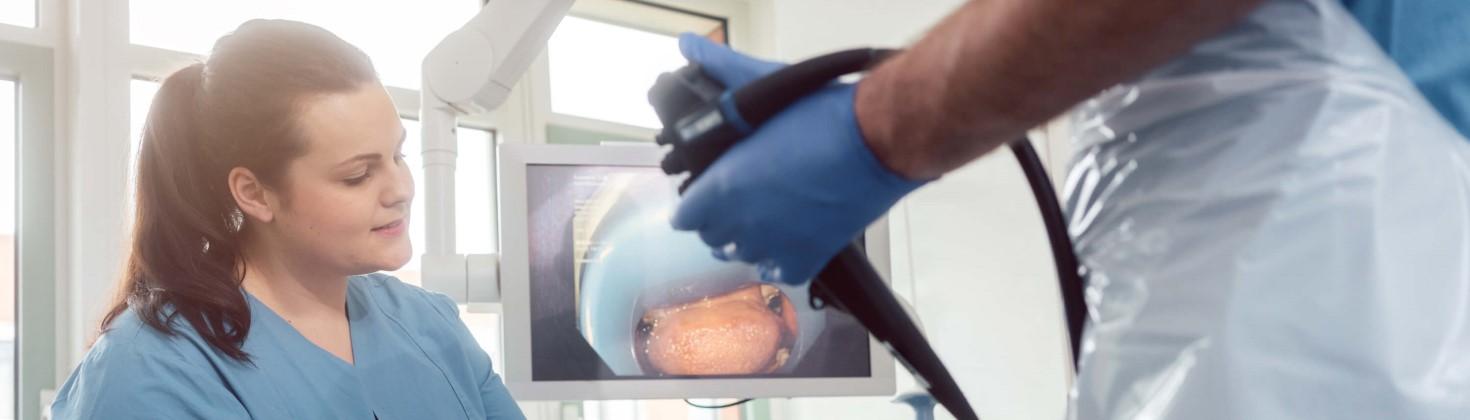
Gastroenterology research taking place at North Bristol NHS Trust
Please speak to the person treating you to see if there is a research study that may be able to help you.
Current Studies:
UK-PSC (A UK Collaborative Study to Determine the Genetic Basis of Primary Sclerosing Cholangitis)
Primary Sclerosing Cholangitis (PSC), a condition characterised by progressive inflammation, obstruction and fibrosis of the intra- and extra-hepatic bile ducts of the liver, presents typically in the fourth or fifth decades leading often to cirrhosis, end stage liver disease and a need for liver transplantation.
The PSC Genetics Study began in 2008, with the aim of sourcing DNA from over 3000 patients with Primary Sclerosing Cholangitis. To date over 1800 patients have participated in the study. This is a significant step towards our goal of identifying genes of potential significance in the pathogenesis of this condition.
Chief Investigator – Dr Gideon Hirschfield
Principle Investigator - Dr Robert Przemioslo
R&I no: 2145
PBC Genetics Study (Investigation of the Genetic and Molecular Pathogenesis of Primary Biliary Cirrhosis)
The PBC Genetics Study is a national effort to establish a PBC DNA collection consisting of DNA samples from approximately 5000 patients with PBC. The DNA collection has already been used for a genome-wide association study (GWAS) of PBC. The next major analysis will be a genome wide association study of response to Ursodeoxycholic acid (UDCA).
Chief Investigator - Dr Richard Sandford
Principle Investigator – Dr Robert Przemioslo
R&I no: 2030
Promise
The The Promise study is: A PROspective randomised double-blind parallel group placebo-controlled multicentre trial of faecal MIcrobiota tranSplantation to improve the primary outcomE (first hospitalisation due to infection) in patients with cirrhosis over 24 months.
End-stage chronic liver disease, known as cirrhosis (irreversible scarring of the liver), is the third biggest cause of mortality and loss of working life in the UK. The human body contains trillions of bacteria, but people with cirrhosis have an increased number of ‘bad’ bacteria in the bowel which makes them highly susceptible to a host of infections. Over-prescribing antibiotics means they are becoming less effective to treat these infections, and the bowel can become infected with ‘super-bugs’. Replacing the unfriendly bowel bacteria in patients with cirrhosis with healthy bacteria donated from healthy volunteers (Faecal Microbiota Transplantation) could be highly beneficial and reduce antibiotic usage.
Project Details
Chief Investigator - Deborah Shawcross
Local PI – Dr Zeino Zeino
R&D 5230
Reduce-2
Palliative Long-term Abdominal Drains Versus Repeated Drainage in Untreatable Ascites Due to Advanced Cirrhosis: A Randomised Controlled Trial (REDUCe 2 Study)
Our aim is to see if palliative long-term abdominal drains (LTADs) result in better quality of life in patients with fluid in the belly due to liver scarring (cirrhosis), compared with current standard of care. The liver can be damaged by excessive alcohol and viral infections. If liver damage continues this scarring leads to permanent damage. It causes a painful buildup of fluid in the abdomen (ascites). Drugs can treat this, but may stop working, leading to untreatable ascites. A liver transplant is then the best option. However, many do not receive a transplant due to concerns about their alcohol use or lack of donors. Life expectancy is on average six months and palliative care is needed. Current palliative care for untreatable ascites involves coming into hospital for 1-2 days, putting a thin tube into the abdomen for a few hours and draining the fluid. This reduces pain from ascites. However, as the ascites builds up quickly, hospital visits are needed every 10-14 days. For people with untreatable ascites due to cancer, palliative care involves placing another tube, an LTAD, into their abdomen. This tube is fitted in hospital but stays in place for months. Nurses/carers then drain smaller amounts of fluid regularly in the community. LTADs avoid frequent hospital visits and can improve quality of life. LTADs are not routinely offered to people with cirrhosis. We conducted a feasibility study where half the participants received LTADs and half continued with standard hospital drainage. The study went well, with no major issues. We now need a larger study to understand the risks/benefits of palliative LTADs in people with cirrhosis. In this larger study we will measure quality of life for both the participants and their carers. The study has been designed with help of the patients/caregivers who are part of the research team.
Chief Investigator – Prof Sumita Verma
Local PI – Dr Ankur Srivastava
R&D - 5300
Boppp
Beta Blockers or placebo for primary prophylaxis of oesophageal varices (BOPPP Trial). A blinded, multi-centre, clinical effectiveness and cost-effectiveness randomised controlled trial
Cirrhosis or liver scarring is an important healthcare problem in the United Kingdom. There are 60,000 patients living with this disease and about 11,000 people die every year due to complications of cirrhosis. Bleeding from the oesophagus or stomach is one of these complications. Cirrhosis causes pressure changes inside the abdomen and swelling of veins in the oesophagus (called “varices”) which can cause fatal bleeding. Research has proven that large varices can be treated with beta-blockers (a type of anti-hypertensive medication) to reduce the pressure in the veins. The management of small varices is still uncertain. This study aims to discover if beta blockers can be used in this setting. We hypothesize that beta blockers will reduce the risk of bleeding from small varices from 20% to 10% over a period of 3 years, resulting in significant cost savings to the NHS from better patient outcomes. The study will be opened in approximately 25 centres nationally. Patients with small volume varices, identified by routine endoscopy, will be randomised to receive either beta-blockers or a placebo. They will be followed up 6 monthly for a period of 3 years. We will assess for complications of cirrhosis (including bleeding) and medicine related side effects. The varices will be monitored annually by endoscope for the duration of the study. General practitioners will be involved in monitoring patients and adjusting doses. As part of our analysis we will assess the impact of treatment on mortality and quality of life using a combined measure, the Quality Adjusted Life-Year (QALY). We will use a mathematical prediction model to estimate the impact of treatment on costs, mortality and quality of life over a patient’s lifetime; identifying if any increased costs are justified by better outcomes for patients and represent good value for money for the NHS budget.
Chief Investigator – Vishal Patel
Local PI – Dr Zeino Zeino
R&D - 4499
Static
STopping Aminosalicylate Therapy in Inactive Crohn’s Disease (STATIC) Study: A Randomized, Open-label, Non-inferiority Trial
Aminosalicylate (5-ASA) agents have proven effective for inducing and maintaining remission in mild to moderate ulcerative colitis (UC) and thus are commonly used as first-line agents for patients with Crohn's disease (CD) in remission. However, there is uncertainty regarding their effectiveness for CD. In this open-label (where both the researchers and participants know which treatment is being administered), randomized study, participants with CD in remission will be allocated to either continue their 5-ASA therapy or withdraw their 5-ASA. The purpose is to investigate if withdrawal of 5-ASA therapy is not unacceptably less effective than continuing on 5-ASA in maintaining CD remission over a 24-month period. There will be a 2 year follow up period to see if participants develop any flares or complications of their CD, to monitor their quality of life and assess the impact on the cost associated with their assigned treatment. The results of the study may help to improve evidence based treatment guidelines and the approach to patient care. Participants will be randomly assigned by a computer to one of two groups: 1. Group 1: Continuation of 5-ASA treatment 2. Group 2: Discontinuation of 5-ASA treatment At the end of the study, half of the participants will be in Group 1 and half of the participants in Group 2. After participants have been assigned to one of these two study groups, they will continue in that group for the duration of their time in the study. Approximately 1580 participants will be taking part in this study. Participants will be asked to attend hospital visits for a total of 5 times over a two-year period, where they will undergo the following procedures at most or all study visits: Blood taking, Physical Exam(only at first visit), vital signs, medication review, side effects review, urine sample, questionnaires and stool samples.
Chief investigator – Dr Gordon Moran
Local PI – Dr Melanie Lockett
R&D – 4472
Take Part in Research
Become one of the thousands of people taking part in research every day within the NHS.
About Research & Development

Find out more about our research and how we're working to improve patient care.
Contact Research
Research & Development
North Bristol NHS Trust
Level 3, Learning & Research building
Southmead Hospital
Westbury-on-Trym
Bristol, BS10 5NB
Telephone: 0117 4149330
Email: research@nbt.nhs.uk