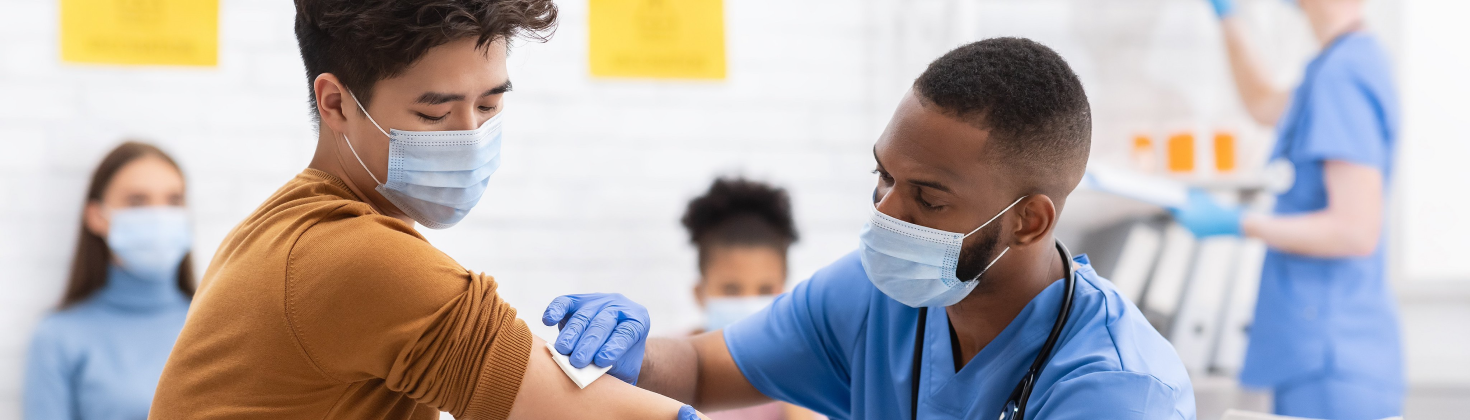
Moderna is conducting a clinical trial, called the mRNA-1273-P305 clinical trial, on investigational booster vaccines that may protect against the COVID-19 Omicron variant and other variants, and you may be eligible to take part.
The mRNA-1273-P305 clinical trial is evaluating the safety and immune response of investigational Omicron variant vaccines compared to a booster dose of Moderna’s COVID-19 vaccine, SPIKEVAX.
This trial is enrolling volunteers 16 years of age or older who are in good or stable health and have received two or three doses of the COVID-19 vaccine at least 90 days ago.
Participation in this trial lasts up to 13 months and includes phone calls and approximately 5-7 scheduled visits to the trial site. Participants will receive a single dose of either one of the investigational Omicron variant vaccines: mRNA-1273.529, mRNA-1273.214, or the already authorized vaccine, SPIKEVAX.
If you are interested in taking part in a vaccine trial, please email us with any of your questions at CovidResearch@nbt.nhs.uk.